Clinical Trials
Clinical Trials for Diabetes, Endocrine, or Metabolic Disorders

Review and Apply for Clinical Trials: -
click the link below:
https://connect.studycatalyst.com/#/listings/93d1fbf7-e97b-410b-bf20-a923fb1f0682
Please contact us for more information on currently enrolling trials.
Some of the trials include:
Diabetes Mellitus Type 1:
Enroll in T1D study
The CATT1 study is a clinical study that is researching the safety and effectiveness of the investigational product at helping manage low blood sugar (hypoglycemia) for type 1 diabetes mellitus patients. Contact us to learn more:

Hypertension and Chronic Kidney Disease Trial: 
Diabetes Mellitus Type 1 Trial involving Once a week basal insulin:
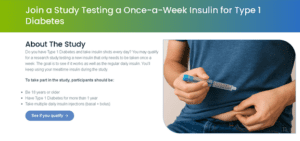
Hyperthyroidism-Grave's disease trial:


Heart Failure Prevention in Diabetics:
Clinical Trial areas include:
Diabetes Mellitus type 2
Diabetes Mellitus Type 1
Obesity
Diabetic complications including CV disease, Chronic kidney disease, Eye disease, peripheral neuropathy
Thyroid disease
Adrenal disorders
Pituitary disease
Metabolic syndrome
Hypertension
Hyperlipidemia
--Please contact our clinical research coordinators – Brandi Kerr, CMA (ext. 106) or Katelyn Glover, BS, MS (ext. 115), or Goknel (Nell) Dumanli, BS (ext. 120), or Rachel Bledsoe, BS (ext. 112), or Teju Gurramkonda, BS, MPH (ext. 116), or Leah Snyder, BS (ext. 117), or Dawn Greer, CMA (ext. 130), or Sydney Anderson, BS (ext. 129), or Ethan Griffith, BA (ext. 125) at (330) 493-0013 to find out if you are eligible for any of the actively recruiting trials at our site.
--You may also let us know of your interest by completing our online appointment request form
or e-mail Brandi Kerr, CRC, Clin. Op. Supervisor at brandik@go2endo.com

If you are wondering what else you can do for your diabetes, endocrine, or metabolic disorder, clinical trials may be just what the doctor ordered: an opportunity to try innovative new devices and treatments not yet available in the marketplace.
A clinical research study is conducted to learn whether a drug, treatment, or method is safe and effective for people who have certain ailments or illnesses. Clinical research studies, also known as clinical trials, follow strict scientific standards to ensure they produce the best data available for health care decision making.
Clinical trials may involve the testing of new:
- Drugs or supplements
- Combinations of existing drugs or supplements
- Ways to deliver existing drugs or supplements
- Medical devices
- Surgical or other techniques
Benefits & Risks
Patients who are interested in participating in clinical trials may be eligible for stipends and transportation assistance. Through your involvement in a clinical trial, you make a valuable contribution to science while helping others who suffer from the same medical condition.
Participants must meet certain criteria, depending on the purpose and scope of the research. Consult Dr. Krishna before participating in any clinical trial to identify the potential risks and benefits to you, based on your particular health condition.

Sponsors
Clinical trials may be funded by public (government) or private entities and may be carried out anywhere in the world. Clinical trials in the US are regulated by the Food and Drug Administration (FDA). In northeast Ohio, Diabetes and Endocrinology Associates of Stark County, Inc. is your local site for diabetes and endocrine-related research.
The multicenter, national and multinational clinical trials conducted at our site have been sponsored by:
- Case Western Reserve University affiliated hospitals
- Cleveland Clinic
- Harvard University affiliated hospitals
- University of Pittsburgh
- Duke University
- Vanderbilt University
- University of Chicago
- Major pharmaceutical companies
- National Institutes of Health (NIH)
- Operation Warp Speed of the US Government
- Google - (Verily-life sciences division)-converging healthcare and cutting-edge technology
- Innovative Biomedical and Biotech startup companies


Reputation and National Recognition:
Our principal investigator and experienced clinical research team members are highly qualified and hold nationally recognized certifications including compliance with Good Clinical Practices (GCP), certification from the Office of Human Research Protections (OHRP) of US Govt. and ethical standards in clinical research.
We have ranked among the top sites nationally and internationally based on our exceptional quality metrics in most major clinical trials. Our site has received commendation certificates for our performance from many sponsors. We have also served as the model site to guide other trial sites in the country.
Some of these landmark trials have been reported in reputed peer reviewed medical journals like the New England Journal of Medicine (NEJM) and our site has been acknowledged in these publications.

Current Clinical Trials
Please contact one of our clinical research coordinators – Brandi Kerr, CMA (ext. 106) or Goknel (Nell) Dumanli, BS (ext. 120), Charlotte Brunkalla, BS (ext. 116), Anika Krishna, MPH, MS (ext. 125), Teju Gurramkonda, MPH (ext. 117), Charlotte Brunkalla, BS (ext. 112), Abby Orellana, BS, MPH (ext. 118), Sydney Anderson, BS (ext. 129), Dawn Greer, CMA (ext. 130)– at (330) 493-0013 to find out if you are eligible for any of the actively recruiting trials at our site.
In addition, April Wallace, BSHCA, CMA (manager), Heidi Daugherty, MSN, CNP (Sub-Principal Investigator) and Dr. Krishna Principal Investigator are also available to answer questions.
You may also let us know of your interest by completing our online appointment request form
or e-mail Brandi Kerr, CRC, Clin. Op. Supervisor at brandik@go2endo.com





Review and Apply for Clinical Trials: -
click the link below:
https://connect.studycatalyst.com/#/listings/93d1fbf7-e97b-410b-bf20-a923fb1f0682
- Call for an appointment today! (330) 493-0013
- Or use online form Request an Appointment





